
Magnesium only slowly reacts with cold water, but rapidly reacts with boiling water or acids. The alkali metals, barium, radium, strontium, and calcium react with cold water. Alkali metals are more reactive than alkaline earths, which in turn are more reactive than transition metals. What’s important is keeping in mind the general trends. Particular metals react more with one acid than another, plus temperature plays a role. The order of the metals in the table comes from experimental data on a metal’s ability to displace hydrogen from water and acid. This is because the conditions of a proposed reaction matter. For example, in some charts, you’ll find sodium listed as more reactive than potassium. If you look around, you’ll notice charts from different sources may order the elements slightly differently. 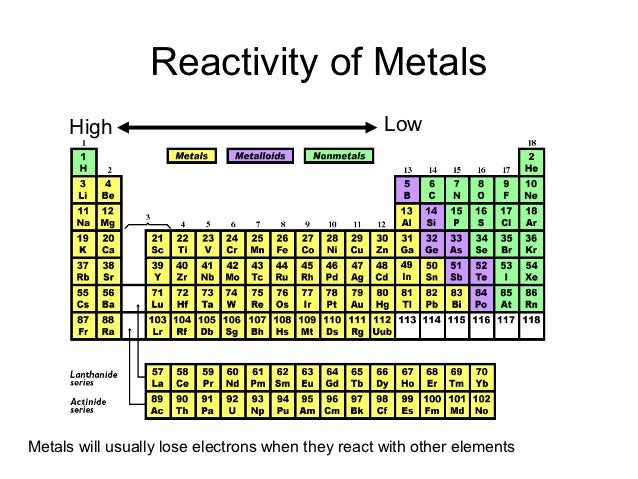
Highly unreactive (Sb reacts with some oxidizing acids) Reacts with acids, generally forming oxides Reacts very slowly with cold water, but vigorously with acids, forming hydroxides Reacts with cold water, replacing hydrogen and forming hydroxide Here is an activity series chart for metals around room temperature. Replacement reactions and ore extraction are two key uses of the activity series. Specifically, use it for identifying whether a metal reacts with water or acid or whether it replaces another metal in a reaction. Knowing the activity series helps you predict whether or not a chemical reaction occurs. 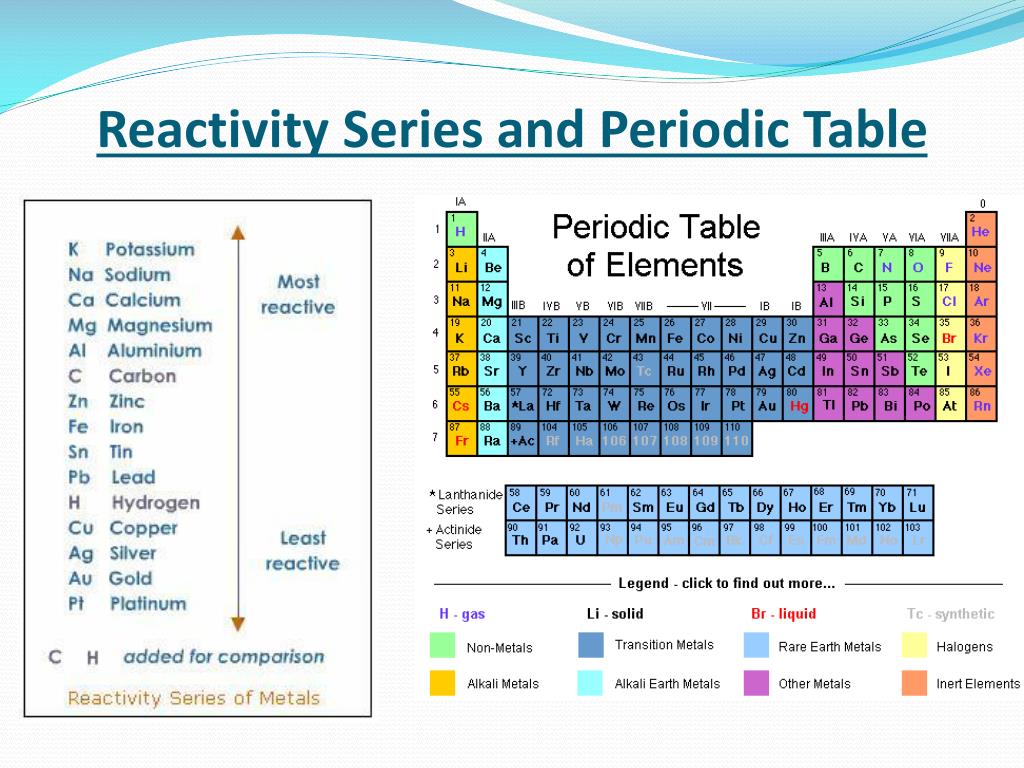
The activity series of metals or reactivity series is a list of metals from most reactive to least reactive. In the reactivity series, the alkali metals are the most reactive, while the noble metals are the least reactive. This entry was posted on Novemby Anne Helmenstine (updated on May 24, 2022)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
